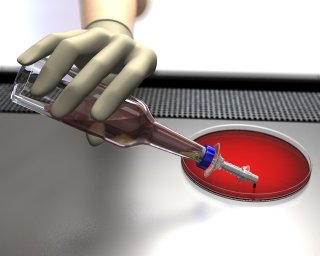
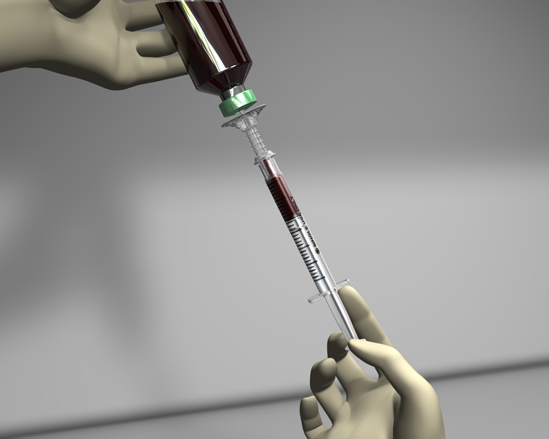
The Safety SubCulture Unit (SCU) is a sharps safety device used to obtain samples from positive blood culture bottles. Needleless sampling reduces risk of needlestick injury.
The plastic tip safely pierces the bottle septum and makes it easy to insert and remove from the culture bottle. The integrated filter cap controls gas or culture discharge for an added level of safety.
Sampling Options
Safety SubCulture Unit offers unique dispensing options for controlled sampling. The SCU sampling channel has been designed to dispense drops directly from a tilted culture bottle.